Mechanisms of Degradation for Polydisulfides: Main Chain Scission, Self-Immolation, Or Chain Transfer Depolymerization
Kristensen, M. M.; Løvschall, K. B.; Zelikin, A. N., Mechanisms of Degradation for Polydisulfides: Main Chain Scission, Self-Immolation, Or Chain Transfer Depolymerization. ACS Macro Lett. 2023, 955-960.
DOI: https://doi.org/10.1021/acsmacrolett.3c00345
Abstract
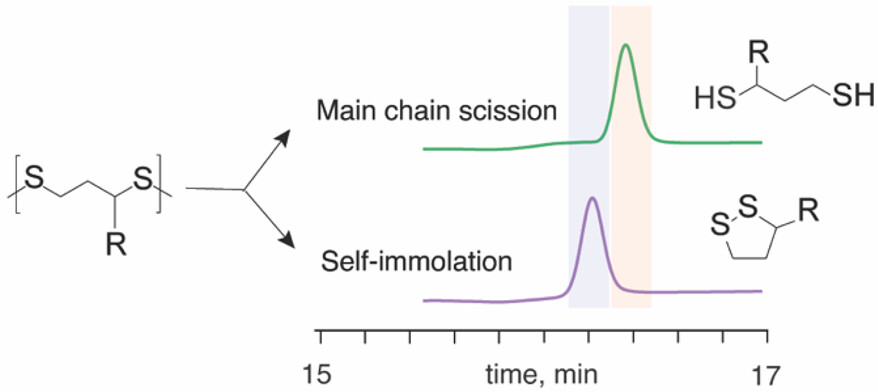
Organic polydisulfides hold immense potential for the design of recyclable materials. Of these, polymers based on lipoic acid are attractive, as they are based on a natural, renewable resource. Herein, we demonstrate that reductive degradation of lipoic acid polydisulfides is a rapid process whereby the quantity of added initiator relative to the polymer content defines the mechanism of polymer degradation, through the main chain scission, self-immolation, or “chain transfer” depolymerization. The latter mechanism is defined as the one during which a thiol group released through the decomposition of one polydisulfide chain initiates depolymerization of the neighbor macromolecule. The chain transfer mechanism afforded the highest yields of recovery of the monomer in its pristine form, and just one molecule of the reducing agent to initiate polymer degradation afforded recovery of over 50% of the monomer. These data are important to facilitate the development of polymer recycling and monomer reuse schemes.