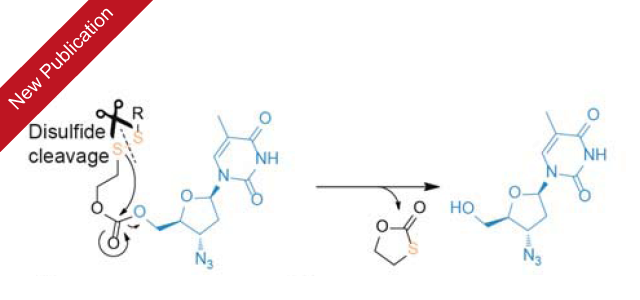
Our newest publication on utilizing self-immolative linker chemistry to construct antiviral macromolecular prodrugs has been published in Chemical Communications.
abstract
The release of azidothymidine from macromolecular prodrugs was designed to respond to the intracellular disulfide reshuffling. This drug has no thiol groups, and a response to this trigger was engineered using a self-immolative linker. The resulting formulations were fast-acting, efficacious, and highly potent with regards to suppressing the infectivity of the virus.